Some strains of gonorrhoea (Neisseria gonorrhoeae; orange) are resistant to several antibiotics.Credit: Science Photo Library
Global map of resistance genes in livestock manure reveals their risk to human health
Livestock farming relies heavily on antibiotics, and manure contains drug-resistant bacteria that can infect people. A study led by researchers at China’s Northwest A&F University in Shaanxi mapped the global patterns of livestock-associated antibiotic-resistance genes that threaten human health.

Nature Outlook: Antimicrobial resistance
The scientists analysed more than 4,000 samples of manure from pigs, chickens and cattle in 26 countries over 14 years. They found more than 3,000 uncharacterized antibiotic-resistance genes, showing that manure hosts a more diverse set of resistant bacteria than was thought.
To assess the potential impact on human health, the researchers calculated each gene’s likelihood of affecting how well antibiotics worked. They took into account the gene’s mobility, the bacterial host’s pathogenicity and the associated antibiotic’s clinical importance.
The health risk of chicken manure exceeded that of pig manure and greatly exceeded that of cattle manure. Chicken manure from African countries was much more likely to contain genes that encode resistance to β-lactam antibiotics, such as penicillin, than was that from other continents. Pig manure from China was more likely to contain resistance genes — especially to tetracycline and aminoglycoside antibiotics such as gentamicin — than was the equivalent from other regions. The biggest driver of risk was the density of livestock production. The researchers hope that their work will help to focus surveillance.
Global clinical trial paves the way for approval of oral gonorrhoea drug
A large clinical trial for treating gonorrhoea has provided the basis for the US approval of an antibiotic last December. The antibiotic, called zoliflodacin (known under the trade name Nuzolvence), will probably be approved in other regions soon, with its developer promising affordable access in low- and middle-income countries.
Gonorrhoea has become increasingly difficult to treat. The sexually transmitted infection can cause infertility if not dealt with, and some strains of Neisseria gonorrhoeae are now resistant to most antibiotics.
The clinical study involved 930 people from five countries with a high gonorrhoea burden: the United States, Belgium, the Netherlands, South Africa and Thailand. It showed that a single oral dose of zoliflodacin treated urogenital gonorrhoea as effectively as did the standard combination of ceftriaxone (given by injection) and oral azithromycin. Zoliflodacin eradicated the infection in more than 90% of cases, without serious side effects.
Zoliflodacin mainly targets an enzyme called DNA gyrase, which is involved in bacterial replication. Other antibiotics also target DNA gyrase, but zoliflodacin avoids the defences of multidrug-resistant strains of N. gonorrhoeae by binding to a distinct part of the enzyme.
Another antibiotic for treating gonorrhoea, gepotidacin (known under the trade name Blujepa), was also approved in the United States last December, after a trial showed that it was as effective as the standard treatment. Gepotidacin not only binds to DNA gyrase, but also targets topoisomerase IV, the bacterial enzyme that untangles DNA molecules after replication.
Large-scale in vitro assay of antibiotic lethality predicts mycobacterial infection outcomes
An in vitro assay that can predict how well antibiotics will work in individuals could pave the way for personalized treatments that reduce the risk of bacteria developing drug resistance.
The method, known as antimicrobial single-cell testing (ASCT), was developed by researchers at the University of Basel in Switzerland and their collaborators. It uses high-content imaging to track, in real time, the behaviour and viability of millions of single cells in response to antibiotic treatments. ASCT captures whether antibiotics kill bacteria, rather than merely inhibiting the microorganisms’ growth — something that has been hard to do at scale.
The team tested how bacteria isolated from 405 people with lung infections caused by Mycobacterium abscessus responded to eight antibiotics. These microbes can sometimes survive long-term antibiotic treatment by entering into a dormant state. Therefore, knowing which antibiotics kill the bacteria outright, and in which people, would be helpful.
The bacteria showed variable responses, suggesting that the specific strain that a person is infected with shapes their treatment response. People with persistent infections often had strains that could tolerate antibiotic treatment. Further study revealed the strains’ genetic characteristics, linked to the antibiotic’s target, that affected drug tolerance.
The authors think that using ASCT to analyse the characteristics of individual infections could lead to the development of personalized therapies that improve outcomes.
Nature Microbiol. 11, 566–583 (2026)
Generative AI designs antibiotics with fresh structures
Scientists at the Massachusetts Institute of Technology in Cambridge and their collaborators have used artificial-intelligence tools to design an antibiotic that combats methicillin-resistant Staphylococcus aureus (MRSA) in mice. The compound has a chemical structure distinct from those of existing antibiotics that could enable it to overcome existing forms of resistance.
The researchers used two generative AI algorithms. The first generates compounds by adding, replacing or deleting chemical components in a starting molecule using information about how atoms combine in bioactive molecules. The second changes a starting molecule on the basis of how bioactive molecules are usually modified. The starting molecules given to the algorithms were ammonia, methane or water.
The algorithms generated 29 million compounds with predicted antibacterial activity. The scientists then shortlisted 90 compounds on the basis of favourable properties such as drug-likeness, ease of synthesis, low toxicity and high antibacterial activity against S. aureus. Of these, 24 were synthesized and tested against S. aureus and other bacteria. One compound, named DN1, showed broad-spectrum activity against Gram-positive bacteria in vitro and cleared MRSA skin infections in mice.
The scientists don’t fully understand how DN1 works, but they think it has several targets, including bacterial-cell membranes. They are now working with Phare Bio, a non-profit social venture, to run further tests on several more-potent analogues of DN1.
Powerful antibiotic discovered in an old biosynthetic pathway
Scientists studying bacteria that naturally produce a well-known antibiotic have unexpectedly found an even more potent compound that is effective against multidrug-resistant bacteria in the laboratory.
Bacteria make compounds that kill competing microbes to aid their own survival, and these chemicals have long been a source of antibiotics. Scientists typically look at the end product of the biosynthetic pathway, because they assume that this compound will be the evolutionarily optimized molecule. But researchers at the University of Warwick, UK, made a different call.
They were studying the pathway that Streptomyces coelicolor uses to make methylenomycin A, an antibiotic identified more than 60 years ago. The team deleted the genes encoding enzymes in this pathway one at a time, and analysed the antimicrobial activity of the molecules produced at each step.
Surprisingly, one of these intermediate compounds was as much as 500 times more effective against several Gram-positive bacteria than was methylenomycin A. The molecule, named premethylenomycin C lactone, was active against methicillin-resistant Staphylococcus aureus, which infects the skin and blood, and Enterococcus faecium, which can cause urinary tract infections and even septicaemia, and is resistant to several classes of antibiotics.
The compound killed two E. faecium strains at around one-thirtieth to one-sixtieth of the amount needed for vancomycin, a last-resort antibiotic, to do so. Moreover, whereas E. faecium developed resistance to vancomycin, this was not seen for premethylenomycin C lactone.
The researchers’ next steps are to work out the compound’s mechanism of action and to see whether fine-tuning its chemical structure can remove any toxic side effects while retaining antibacterial activity.
J. Am. Chem. Soc. 147, 40554−40561 (2025)
Monoclonal antibody therapies can protect against severe multidrug-resistant infections
Monoclonal antibodies are established treatments for cancer and autoimmune diseases. A study now shows that such antibodies can both treat multidrug-resistant infections and protect against them.
Researchers at the Toscana Life Sciences Foundation in Siena, Italy, and their collaborators set out to develop monoclonal antibodies against a multidrug-resistant strain of Klebsiella pneumoniae, a bacterium that can cause pneumonia and infections in the blood and at surgical wound sites. This strain, called ST147, has a high mortality rate and is resistant to numerous antibiotics, including cefiderocol, a next-generation treatment that was introduced only about six years ago.
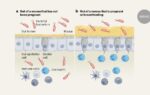
To find antibodies against the ST147 strain, the team obtained blood samples from seven people who had recovered from infection with it. The researchers isolated more than 200 antibodies that bound to antigens on the surface of K. pneumoniae. These antibodies were then cloned, and those that killed the ST147 strain in vitro were studied further.
The most potent antibodies targeted antigens on the microorganism’s outer coat capsule and made the bacterium recognizable to immune cells. The molecules also caused the bacteria to grow in a chain-like structure that promoted immune clearance.
Several antibodies were tested in mice with a blood infection that is fatal in more than 90% of animals. One antibody improved survival to 40% when given one day before ST147 infection, and to around 25% when given one hour after infection.
The authors note that monoclonal antibodies against specific infections can be developed quickly, making them a potentially useful tool for tackling emerging pathogens.