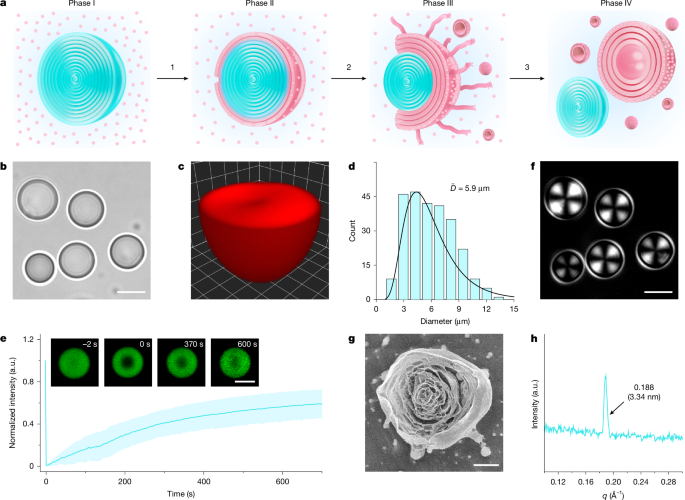
Mu, W. et al. Superstructural ordering in self-sorting coacervate-based protocell networks. Nat. Chem. 16, 158–167 (2024).
Fraccia, T. P. & Jia, T. Z. Liquid crystal coacervates composed of short double-stranded DNA and cationic peptides. ACS Nano 14, 15071–15082 (2020).
Fraccia, T. P. & Martin, N. Non-enzymatic oligonucleotide ligation in coacervate protocells sustains compartment-content coupling. Nat. Commun. 14, 2606 (2023).
Smokers, I. B. A., Visser, B. S., Slootbeek, A. D., Huck, W. T. S. & Spruijt, E. How droplets can accelerate reactions─coacervate protocells as catalytic microcompartments. Acc. Chem. Res. 57, 1885–1895 (2024).
Xu, C., Martin, N., Li, M. & Mann, S. Living material assembly of bacteriogenic protocells. Nature 609, 1029–1037 (2022).
Lu, T. & Spruijt, E. Multiphase complex coacervate droplets. J. Am. Chem. Soc. 142, 2905–2914 (2020).
Ji, Y., Lin, Y. & Qiao, Y. Plant cell-inspired membranization of coacervate protocells with a structured polysaccharide layer. J. Am. Chem. Soc. 145, 12576–12585 (2023).
Cook, A. B., Novosedlik, S. & van Hest, J. C. M. Complex coacervate materials as artificial cells. Acc. Mater. Res. 4, 287–298 (2023).
Gao, N. & Mann, S. Membranized coacervate microdroplets: from versatile protocell models to cytomimetic materials. Acc. Chem. Res. 56, 297–307 (2023).
Ji, Y., Lin, Y. & Qiao, Y. Interfacial assembly of biomimetic MOF-based porous membranes on coacervates to build complex protocells and prototissues. Nat. Chem. 17, 986–996 (2025).
Daly, M. L. et al. Designer peptide–DNA cytoskeletons regulate the function of synthetic cells. Nat. Chem. 16, 1229–1239 (2024).
Dupin, A. & Simmel, F. C. Signalling and differentiation in emulsion-based multi-compartmentalized in vitro gene circuits. Nat. Chem. 11, 32–39 (2019).
Bae, J., Russell, T. P. & Hayward, R. C. Osmotically driven formation of double emulsions stabilized by amphiphilic block copolymers. Angew. Chem. Int. Ed. 53, 8240–8245 (2014).
Rodríguez-Arco, L., Li, M. & Mann, S. Phagocytosis-inspired behaviour in synthetic protocell communities of compartmentalized colloidal objects. Nat. Mater. 16, 857–863 (2017).
Qiao, Y., Li, M., Booth, R. & Mann, S. Predatory behaviour in synthetic protocell communities. Nat. Chem. 9, 110–119 (2016).
Huang, X. et al. Interfacial assembly of protein–polymer nano-conjugates into stimulus-responsive biomimetic protocells. Nat. Commun. 4, 2239 (2013).
Cameron, T. A. & Margolin, W. Insights into the assembly and regulation of the bacterial divisome. Nat. Rev. Microbiol. 22, 33–45 (2024).
Deshpande, S., Spoelstra, W. K., van Doorn, M., Kerssemakers, J. & Dekker, C. Mechanical division of cell-sized liposomes. ACS Nano 12, 2560–2568 (2018).
Holló, G., Miele, Y., Rossi, F. & Lagzi, I. Shape changes and budding of giant vesicles induced by an internal chemical trigger: an interplay between osmosis and pH change. Phys. Chem. Chem. Phys. 23, 4262–4270 (2021).
Ramirez-Diaz, D. A. et al. FtsZ induces membrane deformations via torsional stress upon GTP hydrolysis. Nat. Commun. 12, 3310 (2021).
Dreher, Y., Jahnke, K., Schröter, M. & Göpfrich, K. Light-triggered cargo loading and division of DNA-containing giant unilamellar lipid vesicles. Nano Lett. 21, 5952–5957 (2021).
Zambrano, P. et al. Chemically driven division of protocells by membrane budding. J. Am. Chem. Soc. 146, 33359–33367 (2024).
Zhu, T. F., Adamala, K., Zhang, N. & Szostak, J. W. Photochemically driven redox chemistry induces protocell membrane pearling and division. Proc. Natl Acad. Sci. USA 109, 9828–9832 (2012).
Miele, Y. et al. Self-division of giant vesicles driven by an internal enzymatic reaction. Chem. Sci. 11, 3228–3235 (2020).
Steinkühler, J. et al. Controlled division of cell-sized vesicles by low densities of membrane-bound proteins. Nat. Commun. 11, 905 (2020).
Kurihara, K. et al. Self-reproduction of supramolecular giant vesicles combined with the amplification of encapsulated DNA. Nat. Chem. 3, 775–781 (2011).
Slootbeek, A. D., van Haren, M. H. I., Smokers, I. B. A. & Spruijt, E. Growth, replication and division enable evolution of coacervate protocells. Chem. Commun. 58, 11183–11200 (2022).
Wenisch, M. et al. Toward synthetic life—emergence, growth, creation of offspring, decay, and rescue of fuel-dependent synthetic cells. Chem. 11, 102578 (2025).
Ianeselli, A. et al. Non-equilibrium conditions inside rock pores drive fission, maintenance and selection of coacervate protocells. Nat. Chem. 14, 32–39 (2022).
Song, Y. et al. Budding-like division of all-aqueous emulsion droplets modulated by networks of protein nanofibrils. Nat. Commun. 9, 2110 (2018).
Maruyama, T., Gong, J. & Takinoue, M. Temporally controlled multistep division of DNA droplets for dynamic artificial cells. Nat. Commun. 15, 7397 (2024).
Andes-Koback, M. & Keating, C. D. Complete budding and asymmetric division of primitive model cells to produce daughter vesicles with different interior and membrane compositions. J. Am. Chem. Soc. 133, 9545–9555 (2011).
Dreher, Y., Jahnke, K., Bobkova, E., Spatz, J. P. & Göpfrich, K. Division and regrowth of phase-separated giant unilamellar vesicles. Angew. Chem. Int. Ed. 60, 10661–10669 (2021).
Iwashita, Y. & Tanaka, H. Spontaneous onion-structure formation from planar lamellar nuclei. Phys. Rev. Lett. 98, 145703 (2007).
Izaki, Y., Kurita, R. & Tanaka, H. Hidden linear defects in surfactant onions revealed by coalescence into lamellar layers. Phys. Rev. Res. 3, 043094 (2021).