Xocova (ensitrelvir) has been approved for COVID-19 prevention in Japan on the basis of promising trial results.Credit: The Yomiuri Shimbun via AP Images/Alamy
An antiviral pill has, for the first time, been shown to prevent COVID-19 in people exposed to the SARS-CoV-2 virus at home, according to trial results published today in the New England Journal of Medicine1.
The drug could be a lifeline for those who still face real danger from the virus, such as care-home residents or transplant recipients on immune-suppressing medication.
The advance arrives years after the peak of the COVID-19 pandemic, so the real-world impact might be felt by only a narrow band of individuals. Still, “as a 78-year-old with comorbidities, I certainly would use it if I had a known exposure”, says study co-author Frederick Hayden, a clinical virologist at the University of Virginia School of Medicine in Charlottesville.
Antiviral antidote
The drug, called ensitrelvir, is made by the Japanese pharmaceutical company Shionogi. It blocks an enzyme that coronaviruses need to make new copies of themselves, hitting the same target as one of the two active ingredients in Pfizer’s antiviral Paxlovid. But whereas that ingredient, nirmatrelvir, failed to prevent household infections in trials2, ensitrelvir has come through.
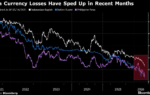
In an international study of more than 2,000 household contacts conducted from June 2023 to September 2024, about 9% of people who got a placebo within 72 hours of a housemate developing symptoms became symptomatic themselves, compared with only about 3% of those who got a five-day course of ensitrelvir. Rates of viral transmission were lower in the ensitrelvir group, too: confirmed infections, symptomatic or not, turned up in only 14.0% of those who received the drug, compared with 21.5% of those who got a placebo.
The therapy generally proved tolerable, without the taste-related side effects often seen with Paxlovid and, overall, similar toxicity as the placebo.
Japan’s health ministry approved ensitrelvir, marketed as Xocova, in March for post-exposure use on the strength of these trial data. Regulators in the United States, Europe and elsewhere are considering whether to follow suit, with a US decision expected in the next month or so.
Long-sought success
The success of ensitrelvir lands in a field marked by repeated setbacks. Paxlovid is the closest a pill has come to demonstrating post-exposure protection, cutting infections by roughly 30% compared with placebo in a household-prevention trial. But the effect fell short of statistical significance.
Other antivirals and would-be preventive drugs have likewise come up short, and antibody infusions that once worked impressively to head off infections after close encounters eventually fared no better, as the Omicron variant and its descendants outflanked them one by one.
Into the void steps ensitrelvir. Yet, now that most people have built up immunity to the coronavirus, either through vaccination or past infection, and with circulating variants now tending to cause milder illness, clinicians continue to debate who, exactly, should be lining up for the prophylactic medication.