
Role of US officials killed in crash in Mexico under scrutiny
The two died in a car crash after a Mexican-led operation to destroy a clandestine drug lab. Source link

The two died in a car crash after a Mexican-led operation to destroy a clandestine drug lab. Source link

Former President Obama is making the case for diplomacy with Iran amid the ongoing conflict, pointing to the 2015 nuclear deal as evidence that Tehran’s program can be constrained without further military intervention. “We went about trying to negotiate a diplomatic agreement that would get the enriched uranium out of Iran, that would assure they……

On the day the Scottish FA announced, external football is now the number one sport in Scotland among girls and women, with participation levels rising by 23% in the past two years – overtaking tennis for the first time – the national team boss stressed the importance to capitalise on the momentum across all levels…

Lisa Kudrow walked onto The Tonight Show Starring Jimmy Fallon this week and did something nobody saw coming. She changed her mind about one of the biggest questions in Friends history. For years, she stayed careful about the whole Ross-and-Rachel break situation. Then Phoebe Buffay herself finally picked a side! The Tonight Show’s Instagram account…

Canadian Prime Minister Mark Carney and Finnish President Alexander Stubb hit the ice alongside the Ottawa Charge hockey team for a friendly match. The event took place during Stubb’s first formal bilateral visit to Canada. According to an official statement, the visit aims to deepen cooperation between Finland and Canada across trade, defense, technology, energy,…

It’s an objective fact that humanity’s footprint on the planet cannot be erased—we will affect the land and seas and air one way or another through technology, society, and certainly travel. In the past, the goal was to mitigate or lessen the affects we and the tourism industry have on the earth. Now, the point…
Prices, debt, and scarcity will strike a blow against the world’s workshop Source link

In the face of a devastating US-Israeli bombing campaign, Iran’s rival political factions cast aside their differences to unite behind the country’s decision-makers. But since a cease-fire with the United States came into force on April 8, deep-rooted political differences in the Islamic republic have spilled out into the open. The public rupture over Tehran’s…
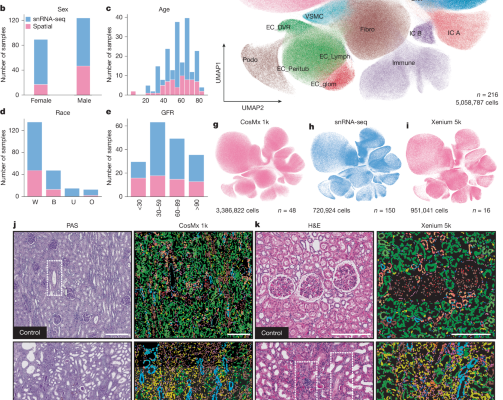
Sample acquisition and ethics approval The University of Pennsylvania institutional review board (IRB) approved the collection of human kidney tissue for this study. Left-over kidney samples were irreversibly de-identified, and no personal identifiers were gathered. Therefore, they were exempt from IRB review (category 4). We engaged an external, honest broker who was responsible for clinical…

Discord is announcing the launch of Nitro Rewards today, a new program for subscribers that bundles benefits from third parties like Microsoft, Logitech, and SteelSeries, into a Nitro subscription. The main addition is that Nitro members will now get a new starter edition of Xbox Game Pass at no extra cost. Xbox Game Pass starter…