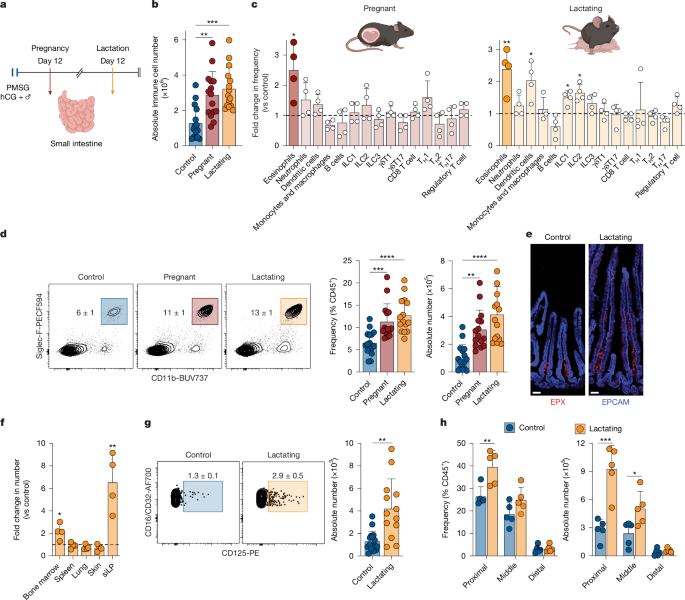
Mice
All animal experiment were performed in accordance with protocol approved by the Institutional Animal Care and Use Committee and Institutional Biosafety Committee at Princeton University. C57BL/6 (stock no. 000664), Lgr5eGFP−IRES-creERT2 (stock no. 008875), BALB/cJ (stock no. 000651) and ΔdblGATA (stock no. 033551) mice were purchased from The Jackson Laboratory. Germ-free C57BL/6 mice were purchased from Taconic Biosciences. For timed pregnancy, females were intraperitoneally injected with 2 IU PMSG followed by 2 IU hCG 46–48 h later and then co-housed with male breeder mice. This hormone-induced timed pregnancy model was used for all experiments unless otherwise specified. For all experiments, C57BL/6 male breeder mice were used unless otherwise specified. Tissue collection and analyses were performed at embryonic day 12.5 for pregnant mice or 12 days postpartum for lactating mice unless otherwise specified. For long-term experiments, pups were weaned at 3 weeks and female mice were maintained for 12 weeks post-lactation. Age-matched mice that were administrated with hormones and co-housed with male mice but did not get pregnant were used in each experiment as nulliparous controls. Animals were randomly assigned to experimental groups whenever possible, and sample processing and outcome assessment were performed in a blinded manner where feasible.
Bacteria and infections
Wild-type Y. pseudotuberculosis IP32777 was grown overnight in 2× YT medium (Sigma-Aldrich) at 25 °C with shaking at 200 rpm. A nalidixic-acid-resistant strain of S. Typhimurium, IR715, was cultured in LB at 37 °C with shaking at 200 rpm. L. monocytogenes strain 10403s was cultured overnight in brain heat infusion broth (Teknova) at 37 °C with shaking at 200 rpm. Overnight cultures were centrifuged at 4,000g, resuspended in PBS and adjusted to a density (absorbance at 600 nm) of 1.0 in PBS. Mice fasted overnight (12–16 h) in new cages were orally administered 200 μl of the indicated pathogen suspension. To determine bacterial burden in the maternal spleen and faecal samples, spleens were homogenized through 70 μm filters with PBS, whereas faecal samples were collected in pre-weighed tubes and homogenized in PBS. For Y. pseudotuberculosis, homogenates were serially diluted and plated onto Yersinia-selective agar with Yersinia-selective supplement (Millipore), and colonies were counted after incubation at 25 °C for 48 h. For S. Typhimurium, homogenates were serially diluted and plated onto LB agar with 50 mg ml–1 nalidixic acid and incubated overnight at 37 °C. For L. monocytogenes, homogenates were serially diluted and plated onto brain heart infusion agar (Sigma Aldrich) with 100 mg ml–1 streptomycin and incubated overnight at 37 °C. All lactating dams were infected between day 5 and 8 postpartum period.
Tissue processing
Mice were euthanized with CO2. Tissues (small intestine, bone marrow, lung, skin and spleen) were collected into complete medium (RPMI 1640 supplemented with 20 mM HEPES, 2 mM l-glutamine, 1 mM sodium pyruvate, 1X nonessential amino acids, 55 μM β-mercaptoethanol, 100 U ml−1 penicillin and 100 μg ml–1 streptomycin) with 3% FBS on ice until further processing. For compartmentalization analysis, we defined the first 10 cm adjacent to the stomach as proximal, the last 10 cm as distal and the intervening segment as middle. For cell isolation from the small intestinal lamina propria, Peyer’s patches were removed and tissues were opened longitudinally, washed with cold PBS twice, cut into 1 cm segments and placed immediately into 3% complete medium containing 5 mM EDTA and 0.154 mg ml−1 (1 mM) dithiothreitol. Tissues were then incubated for 20 min at 37 °C with continuous stirring at 400 rpm, vigorously shaken in RPMI containing 20 mM HEPES, 2 mM EDTA, 100 U ml–1 penicillin and 100 μg ml–1 streptomycin for 1 min 3 times to remove any remaining mucus and further digested with 10 ml of 0% medium containing 0.5 mg ml–1 DNase I (Sigma-Aldrich) and 0.1 mg ml–1 Liberase TL (Roche) for 25 min at 37 °C with constant stirring at 400 rpm. Lungs were diced and incubated in 2 ml of prewarmed medium containing 0.5 mg ml−1 DNase I and 0.1 mg ml–1 collagenase IV for 35 min at 37 °C. To isolate cells from the skin, ear pinnae were excised and separated into ventral and dorsal sheets, digested by placing the dermal side down in medium containing 0.5 mg ml–1 DNase I with 0.25 mg ml−1 Liberase TL and incubated for 90 min at 37 °C. Digested intestines, lung and skin were passed through 70 μm cell strainers and centrifuged at 1,500 rpm for 5 min. Leukocytes were enriched by resuspending the samples in 4 ml of 37.5% Percoll and centrifuged at 1,800 rpm for 5 min. For blood analysis, 200 μl blood was added with 1 ml of ACK lysing buffer and incubate on ice for 10 min with occasional shaking. The reaction was then quenched with 5 ml of 3% medium followed by spinning at 1,800 rpm for 5 min. The pellet was then resuspended in 3% medium. Cells were washed with HBSS once before downstream staining and analyses.
Flow cytometry analysis
Cells isolated from tissues were plated into 96-well plates with counting beads. To measure cytokine production potential, cells were re-stimulated in complete IMDM supplemented with 10% FBS, 50 ng ml–1 PMA, 5 μg ml–1 ionomycin and 3 μg ml–1 BFA for 2.5 h at 37 °C. Cells were washed twice with cold HBSS before staining. Surface staining was performed on ice for 20 min, followed by washing with HBSS once and 10 min of Live/Dead staining. Cells were then washed twice with HBSS and fixed for 1 h at room temperature using either eBioscience intracellular fixation buffer (00-5123-43, Invitrogen) if intracellular staining was needed or BD Cytofix Fixation buffer (554722, BD Biosciences) for surface staining only. For intracellular staining, fixed cells were washed twice with the corresponding permeabilization buffer and intracellular staining was performed overnight at 4 °C. Before flow analysis, cells were washed twice and resuspended in 200 μl cold PBS. Flow cytometry analysis was performed on a FACSymphony A3.
The following antibodies were used for flow cytometry: anti-CD90-2 (FITC; 1:200; 53-2.1), anti-CD90-2 (BV786; 1:400;3 0-H12), anti-MHCII (BV650; 1:500; M5/114.15.2), anti-CD64 (BV605; 1:100; X54-5/7.1), anti-CD103 (BV510; 1:100; 2E7), anti-Ly6C (APCCy7; 1:200; HK1.4), anti-F4/80 (AF700; 1:200; BM8), anti-PD-L1 (PE; 1:100; 10 F.9G2), anti-TCRγ/δ (BV650; 1:200; GL3), anti-IFNγ (BV605; 1:200; XMG1.2), anti-CD4 (BV510; 1:200; RM4-5), anti-IL17A (BV421; 1:200; TC11-18H10.1), anti-T-bet (PECy7; 1:200; 4B10), anti-CD48 (BV786; 1:200; HM48-1), anti-CD45R/B220 (BV421; 1:400; RA3-6B2), anti-CD135 (APC; 1:200; A2F10), anti-CD150 (PECF594; 1:200; TC15-12F12.2), anti-FcεRIα (AF700; 1:200; MAR-1), anti-CCR3 (PECy7; 1:200; J073E5), anti-cKit (PECy7; 1:200; 2B8) and anti-CD200R3 (PE; 1:100; Ba13) were from BioLegend; anti-CD11c (BV786; 1:200; N418), anti-B220 (BUV805/PerCPe710; 1:200; RA3-6B2), anti-CD11b (BUV737; 1:100; M1/70), anti-CD45 (BUV395/APCCy7; 1:200/1,000; 30-F11), anti-FOXP3 (FITC; 1:400; FJK-16s), anti-TCRb (BUV737; 1:100; H57-597), anti-CD44 (AF700; 1:100; IM7), anti-GATA3 (APC; 1:200; TWAJ), anti-IL-5 (PE; 1:200; TRFK5), anti-IL-13 (PE; 1:200; eBio13A), anti-cKit (PerCPe710; 1:200; 2B8), anti-CD34 (AF488; 1:100; RAM34), anti-SCA1 (BV605; 1:400; D7), anti-CD16/CD32 (AF700; 1:200; 93) and anti-CD127 (PECy7; 1:200; A7R34) were from eBioscience; and anti-Ly6G (BV650/APC; 1:200; 1A8), anti-Siglec-F (PECF594/BV510; 1:200; E50-2440), anti-CD8b (BUV395; 1:400; H35-17.2), anti-RORγt (PECF594; 1:400; Q31-378), anti-CD125 (PE; 1:100; T21) and anti-CD90-2 (BUV805; 1:200; 30-H12) were from BD Bioscience.
Imaging of intestinal tissues
The small intestine was removed from the animal immediately after CO2 euthanasia and the first 10 cm was sectioned. The tissue was flushed with cold PBS using a gavage needle attached to a 20 ml syringe to remove all faeces and excess liquid was removed from inside. The tissue was dangled over a fix tray and 3 ml of fix (1 volume of Cytofix in 2 volumes of PBS) was flushed through the tissue. The tissue was kept on ice for 1 h and then laid straight on a blotting paper soaked with cold PBS. An L-shaped scissor was used to cut the tissue open lengthwise. The two ends of the tissue were sliced off, and the remaining tissue was rolled into a tight spiral with the distal end inside. The roll was then pierced with a 19 G needle and fixed with 2 ml cold fix in a 14-ml Falcon tube overnight at 4 °C. The next day, the roll was transferred to 30% sucrose in PBS and stored overnight at 4 °C. For embedding, the roll was transferred into a 14-ml Falcon tube with OCT compound and shaken at 200 rpm at room temperature for 4–8 h. The tissues were then removed from the needle and placed into moulds filled with OCT so that the entire tissue was covered with OCT and the upward face was a spiral. The mould was placed on a pre-chilled metal tray on dry ice to freeze immediately. After complete freezing, the blocks were kept at –80 °C for long-term storage. Sections were cut at 20 μm using a cryostat at –20 °C and stored at –80 °C for at least 24 h before staining.
For immunofluorescence staining, slides were equilibrated to room temperature and fixed in pre-chilled acetone for 10 min at –20 °C. An ImmEdge Hydrophobic Barrier PAP Pen (Vector Laboratory) was used to draw squares around the tissues and the slides were air dried at room temperature for 1 h. The tissues were rehydrated with cold PBS 3 times for 5 min each and blocked with base block buffer (1% BSA and 0.24% Triton X-100 in PBS) for 1 h at room temperature. Primary antibodies were diluted in the base block buffer and staining was performed either for 4 h at room temperature or overnight at 4 °C. The tissue samples were then washed with PBS twice, 5 min each, stained with DAPI in PBS for 30 min at room temperature and washed again with PBS for 3 times. Excess liquid was carefully removed from the slides using a Kimwipe. The slides were mounted with Prolong Gold Antifade reagent (ThermoFisher) and dried overnight before imaging on a Nikon CSU-W1. For measurement of goblet cell counts and villus and crypt lengths, at least ten crypt–villus units were measured for each mouse. For LGR5+ stem cell imaging, Lgr5eGFP-IRES-creERT2 mice were used. For MUC2 staining, 1.5 cm of the proximal and distal small intestine was cut off and put into Carnoy’s fixative at 4 °C overnight. The next day, tissue samples were transferred to 70% ethanol and placed at 4 °C for at least 24 h. Tissue samples were then processed on an ASP300S tissue processor and embedded in paraffin for long-term storage. Slides were sectioned at 7 μm. Slides were deparaffinized and incubated in 1× antigen retrieval buffer at 95 °C for 30 min and washed twice with PBS for 5 min each. Tissues were then permeabilized with 0.1% Triton X-100 in PBS for 10 min and blocked with blocking buffer (3% BSA and 0.1% Tween-20 in PBS) for 1 h at room temperature. Anti-MUC2 primary antibody was diluted in blocking buffer and applied to tissues overnight at 4 °C. The next day, tissue samples were washed with PBS twice, 5 min each, stained with fluorochrome-conjugated secondary antibody and DAPI in blocking buffer for 1 h at room temperature and washed again with PBS for 3 times. Mounting and imaging were performed as described above.
The following antibodies were used for confocal imaging. Anti-mouse antibodies were anti-EPCAM (AF647; 1:1,000; 118212; BioLegend), anti-MUC2[C3] (1:200; GTX100664; Genetex), anti-EPX (MM25-82.2.1; 1:1,000; by E. A. Jacobsen from Mayo Clinic). Other antibodies were anti-GFP (AF488; 1:400; A21311; Invitrogen) and UEA1 (Rhodamine; 1:200; RL-1062; Vector Laboratory). Secondary antibodies were goat anti-rabbit IgG H&L (AF594; 1:500; ab150080; Abcam).
Intestinal epithelial cell sorting
Small intestines were collected from CO2-euthanized animals and the first 10 cm was sectioned. The tissues were opened longitudinally, washed 2 times with 5% gut wash buffer (HBSS supplemented with 25 mM HEPES, 2 mM l-glutamine, 1 mM sodium pyruvate, 1X nonessential amino acids, 100 U ml–1 penicillin and 100 μg ml–1 streptomycin with 5% FBS) and cut into 2–5 mm pieces into 10 ml gut wash buffer containing 2 mM DTT. The tissue was incubated in a 37 °C water bath for 10 min, briefly shaken every 2 min and passed through a 100 μm filter after vigorous shaking 15 times. The tissue was then transferred to a new conical tube containing 10 ml gut wash buffer with 5 mM EDTA and incubated at 37 °C for 8 min with brief shaking every 2 min. The supernatant fraction was collected on ice by passing through a 100 μm filter and the tissue was transferred to a new conical tube to repeat the EDTA wash step for a total of 3 times. The second and third fractions were collected in the same tube as the first one. Cells were centrifuged at 1,000 rpm for 5 min at 4 °C and washed once with 0% DMEM (supplemented the same as gut wash buffer but with different FBS concentration, referred to as DMEM below). The cell pellet was incubated in 10 ml 0% DMEM supplemented with 0.1 U ml–1 Dispase and 0.1 mg ml–1 DNase in a 37 °C water bath for 8 min, with brief shaking every 2 min. After neutralization with 5 ml 2.5% DMEM, the cells were centrifuged at 1,000 rpm for 5 min and washed once with the staining buffer (same as the 5% gut wash buffer). Antibodies diluted in the staining buffer were added and staining was performed on ice for 20 min. The cells were washed with 10 volumes of 10% DMEM and resuspended in 10% DMEM for sorting. All cell sorting was performed on a Sony MA900 sorter. Before loading onto the sorter, the cell suspension was filtered using a 5 ml Falcon tube with a cell strainer cap and DAPI was added at a 1:400 concentration. Alive EPCAM+ (anti-EPCAM (AF594; 1:1,000; G8.8; BioLegend)) epithelial cells were sorted directly into 10% DMEM.
scRNA-seq and analysis
EPCAM+ IECs were sorted from 12–15-week-old nulliparous, pregnant or lactating mice as described in the previous section. For each group, cells were pooled from 3 mice and a total number of 25,000 cells per lane were loaded onto a Chromium Single Cell Controller. Libraries were prepared using Chromium Next GEM Single Cell 3′ Reagent kits v.3.1 (10x Genomics) following the manufacturer’s instructions and sequenced on a NovaSeq 6000 System. Cell by gene count matrices were generated for each sample using the count function from CellRanger v.7.1.0 (10x Genomics).
Data were analysed using Scanpy (v.1.8.2). Count matrices from the three samples were concatenated then filtered and processed together. Data were filtered to keep only cells with at least 10 expressed genes, 100 total counts and less than 15% of total counts coming from mitochondrial genes. Genes were kept if they were expressed in at least 1% of filtered cells (211 cells) and had at least 250 total counts across cells.
For clustering and cell-type assignment, the count matrix was normalized by Pearson residuals (θ = 1) and then used for principal component analysis (100 components), k-nearest neighbour graph construction (k = 30, number of principal components = 50) and Leiden clustering. Clusters were annotated on the basis of the expression of known marker genes. In some cases, clusters were further subclustered and then annotated by cell-type identity. Cells that were potentially doublets (expressing marker genes from multiple cell types) or unknown were removed before downstream analyses.
For differential gene expression, the raw count matrix was normalized by library size and then log-transformed. For each cell type, differential expression was performed between nulliparous versus pregnancy and nulliparous versus lactating conditions. Genes were filtered out if they were not expressed by at least 5% of cells in either condition. For remaining genes, log fold change values were calculated as the difference between the mean expression values of log-library size normalized counts for the compared conditions. P values were calculated using a Mann–Whitney U-test and corrected for multiple hypothesis with the Benjamini–Hochberg procedure.
Spatial transcriptomic analysis using Xenium Prime Mouse 5K
The first 10 cm of the small intestine was collected from mice, fresh-frozen with isopentane and embedded in OCT. Tissues were cryosectioned at 10 μm, mounted on Xenium slides and processed according to the Xenium Prime 5K protocol (10x Genomics). Xenium datasets were generated using a Xenium Analyzer (instrument software v.3.1.0.0 and analysis v.xenium-3.1.0.4). Tissue slides were also stained with Xenium MultiTissue Stain mix for multimodal cell segmentation, and this segmentation from Xenium Analyzer was used for all analyses. Post-Xenium Analyzer haematoxylin and eosin staining was performed according to the official demonstrated protocol.
Transcripts for each cell were filtered to keep only transcripts overlapping the nucleus segmentation for each cell. Cells from all samples were concatenated and the subsequent count matrix was further filtered to keep only cells with counts from at least ten genes and with exactly one segmented nucleus. Data were analysed using Scanpy (v.1.10.4). The count matrix was normalized by library size, log-transformed and then z-scored across genes. Normalized data were used for principal component analysis (50 components), k-nearest neighbour graph construction (k = 15) and Leiden clustering. Each cluster was then separately clustered into more fine-grained clusters. A count matrix for the same cells but for all transcripts, including non-nuclear transcripts, was also generated and normalized as described above. These counts were used to confirm the expression patterns for the nuclear-only transcript counts.
Clusters and subclusters were manually annotated on the basis of the expression of known marker genes. Cluster-specific differentially expressed genes were identified by calculating log fold change values of the mean expression between normalized counts for cells included and excluded in each cluster. For this analysis, counts were normalized by library size only. P values were calculated using a Mann–Whitney U-test and corrected for multiple hypothesis testing with the Benjamini–Hochberg procedure. Myeloid clusters were identified on the basis of Cd45 and Cd11b expression, and eosinophils were further labelled on the basis of Il5ra and Ccr3 expression in the myeloid component. Differentially expressed genes between nulliparous and lactating conditions for eosinophils were identified using DESeq2 with a pseudobulk approach50. 10x Genomics Xenium Explorer (v.3.2.0) was used to visualize cell and transcript location and to generate spatial images of cell-type identity overlaying DAPI staining.
Swiss rolls were computationally unrolled into the proximal-distal axis and normalized crypt–villus axis using a previously described approach51. In brief, for each sample, the base of the small intestine roll was manually annotated, and the nearest location on the base spiral was calculated for each cell. The relative position of this nearest location was used as the proximal–distal position and the distance to this nearest location was used as the crypt–villi position, normalized by local maxima to be between 0 and 1. For plotting kernel density estimates of the distribution of cell counts along the proximal–distal axis and crypt–villus axis, we applied a Gaussian KDE (bandwidth determined using Scott’s rule). The KDE was scaled so that the total area under the curve equals the total cell count using a grid size of 500 bins. An individual KDE was calculated for each sample, and the mean and standard deviation of these KDE curves are shown. Neighbouring cell analysis was performed by identifying the directly adjacent spatial neighbours for each cell by using cell segmentation boundaries within 2 μm of each other.
Organoid culture and imaging
Epithelial cells were isolated and sorted from Lgr5eGFP-IRES-creERT2 mice using a MA900 cell sorter as described in the previous section. Alive EPCAM+LGR5+ cells were gated as shown in Extended Data Fig. 5b and directly sorted into cold complete organoid growth medium (OGM) and centrifuged at 400g for 8 min. The cell pellet was then resuspended in complete OGM at a concentration of 40 cells per μl and mixed with 1 volume of undiluted Matrigel matrix. Next, 10 μl of the mixture was quickly pipetted into the centre of each well of a 96-well plate. The plate was immediately placed into a 37 °C incubator for 10 min for the Matrigel to set, and 100 μl pre-warmed complete OGM supplemented with 10 μM Y-27632 was added to each well. The medium was changed every other day and Y-27632 was only supplemented for the first 3 days. On day 8, plates were centrifuged at 3,000 rpm for 10 min at 10 °C to spin the organoids down. Organoids were then fixed in 4% paraformaldehyde and PBS overnight at 4 °C and washed with PBS 3 times before staining. Organoids were dehydrated with 30% sucrose in PBS for 30 min at room temperature and permeabilized with permeabilization buffer (0.1% Triton X-100 and 3% BSA in PBS) for 1 h at room temperature. Primary staining was performed using the permeabilization buffer either for 4 h at room temperature or overnight at 4 °C. Organoids were washed twice with cold PBS, stained with DAPI for 30 min at room temperature and then washed again with PBS 3 times. Next, 50 μl CytoVista 3D Cell Culture Clearing reagent (Invitrogen) was added to each well and the plate was placed at 4 °C for 10 min before imaging on a Nikon CSU-W1. Clonogenicity was assessed on day 8 by quantifying the percentage of organoid formed out of the number of LGR5+ intestinal stem cell initial seeded.
Eosinophil isolation and ex vivo culture
Intestinal lamina propria was prepared as described in the previous section. Alive CD45+CD11b+Siglec-F+ eosinophils (anti-Siglec-F (PE; 1:200; E50-2440; BD), anti-CD11b (AF647; 1:100; M1/70; BioLegend) and anti-CD45 (APC-eF780; 1:200; 30-F11; eBioscience)) were directly sorted into cold complete RPMI medium supplemented with 20% FBS. Cells were centrifuged at 400g for 8 min and counted so that 30,000 eosinophils were resuspended in 65 μl 20% RPMI and plated in each well of a 96-well plate. Next, 65 μl of 20% RPMI supplemented with 20 ng ml–1 IL-5 was overlaid, and the plate was gently swirled before being placed into an incubator set at 37 °C with 5% CO2. After 48 h, the plate was centrifuged at 1,600 rpm for 5 min at 4 °C. A volume of 120 μl supernatant was collected from each well and stored at –80 °C until analysis. Cytokine and growth factors were measured using multiplex assay kits.
Blockade of eosinophil migration using anti-CCR3 antibody
Timed-pregnant mice were retro-orbitally injected with 1 mg of anti-mouse CCR3 antibody (BE0316; BioXCell) or rat IgG2b isotype control weekly, beginning from gestational day 10.5 until day 10 postpartum for a total of 4 doses and analysed at day 12 postpartum. For infection analysis, mice received a total of 3 doses before the analysis. Small intestine samples were collected and analysed by flow cytometry to confirm successful blockade of eosinophils.
Eosinophil chemotaxis assay using Transwell
Mice were treated with 1 mg isotype or anti-CCR3 antibody. At 24 h after treatment, alive CD45+CD11b+Siglec-F+ eosinophils were isolated from bone marrow and resuspended at a concentration of 2 × 105 cells per ml in 20% RPMI medium without rmIL-5. In vitro chemotaxis assays were performed in HTS Transwell plates (Corning) with a 5 µm pore size. Recombinant mouse eotaxin-1 and eotaxin-2 (R&D) were prepared in a concentration of 100 ng ml–1 using the same medium. For cell migration assays, 100 µl cell suspension was added to the upper well and 100 µl medium with or without eotaxins was added to the bottom well, followed by 1 h of incubation at 37 °C. Migrated eosinophils were collected from the bottom compartment, analysed by flow cytometric counting and normalized to no-eotaxin control.
Morphometric analysis for the mucus layer
For the quantitative analysis of the mucus area present in the duodenum tissue, a previously described method52 was used. PAS and Alcian Blue-stained histological sections were visualized and scanned using a Scan Aperio slide scanner (Leica Biosystems) at ×40 magnification. For each mouse, the entire duodenum mucosa was analysed for the quantification of the mucin area (PAS and Alcian Blue-positive) with the aid of QuPath software (https://qupath.github.io)53. The total length of each section was manually measured in micrometres to determine the actual proportion of the duodenal surface analysed for each animal. Then, the mucus area for each animal was normalized to the respective duodenum mucosa length. The methodology used for imaging and morphometric conditions was adapted from a previously published method54.
Measurement of gut permeability
Intestinal permeability was measured by performing oral gavage of 4 kDa FITC-labelled dextran (Sigma-Aldrich) and measuring fluorescent translocation into the plasma. Mice were fasted for 4 h before being orally gavaged with 200 μl of 60 mg ml–1 (12 mg) FITC-dextran. Two hours later, blood was retro-orbitally collected and centrifuged at room temperature at 12,000g for 15 min to obtain the plasma. Fluorescence intensity was then measured on fluorescence plates using an excitation wavelength of 485 nm and an emission wavelength of 535 nm.
Statistical analysis
Two-tailed unpaired Student’s t-tests, one-way ANOVA and two-way ANOVA were performed to compare the results using GraphPad Prism (v.10). No samples were excluded from analyses. We treated P < 0.05 as significantly different, with *P < 0.05, **P < 0.01, ***P < 0.001 and ****P < 0.0001. Details regarding the exact sample size for each experimental group and condition and representative data are presented in the figure legends.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.